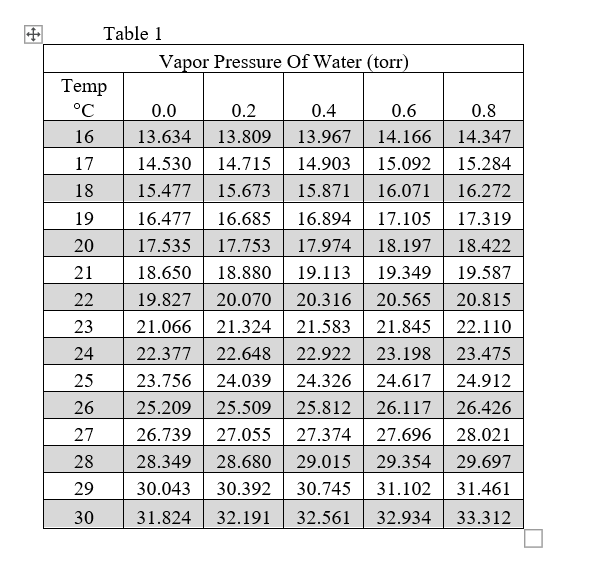
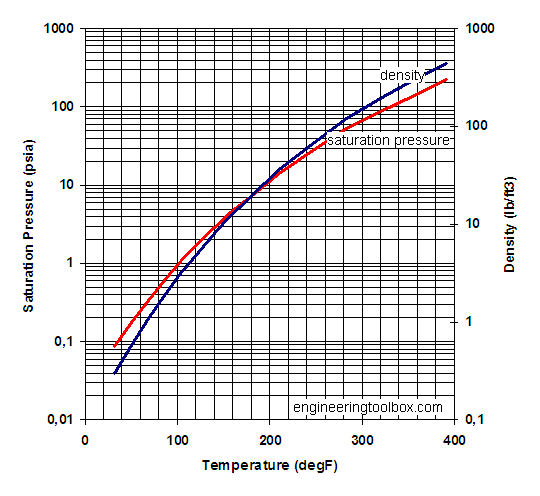
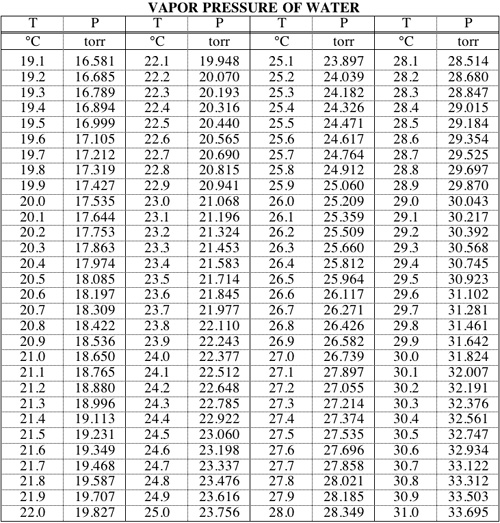
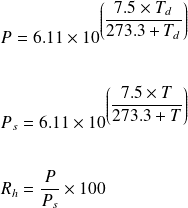Calculate vapour pressure of 0.1M urea soln. Vapour pressure of water at the given temperature is 20 torr. Assume molality and molality to be equal.
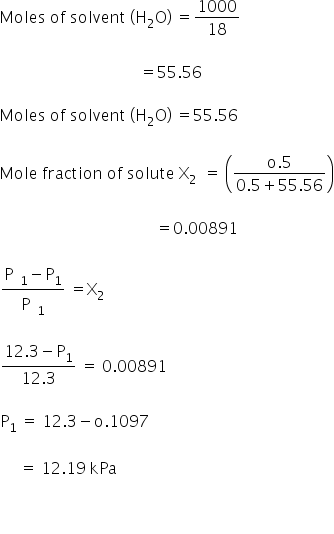
The vapor pressure of water is 12300 Pa at 27 C. Calculate the vapor pressure of 0.5 M solution of a non dissociating/non associating solute in it. - hk3hf2ll